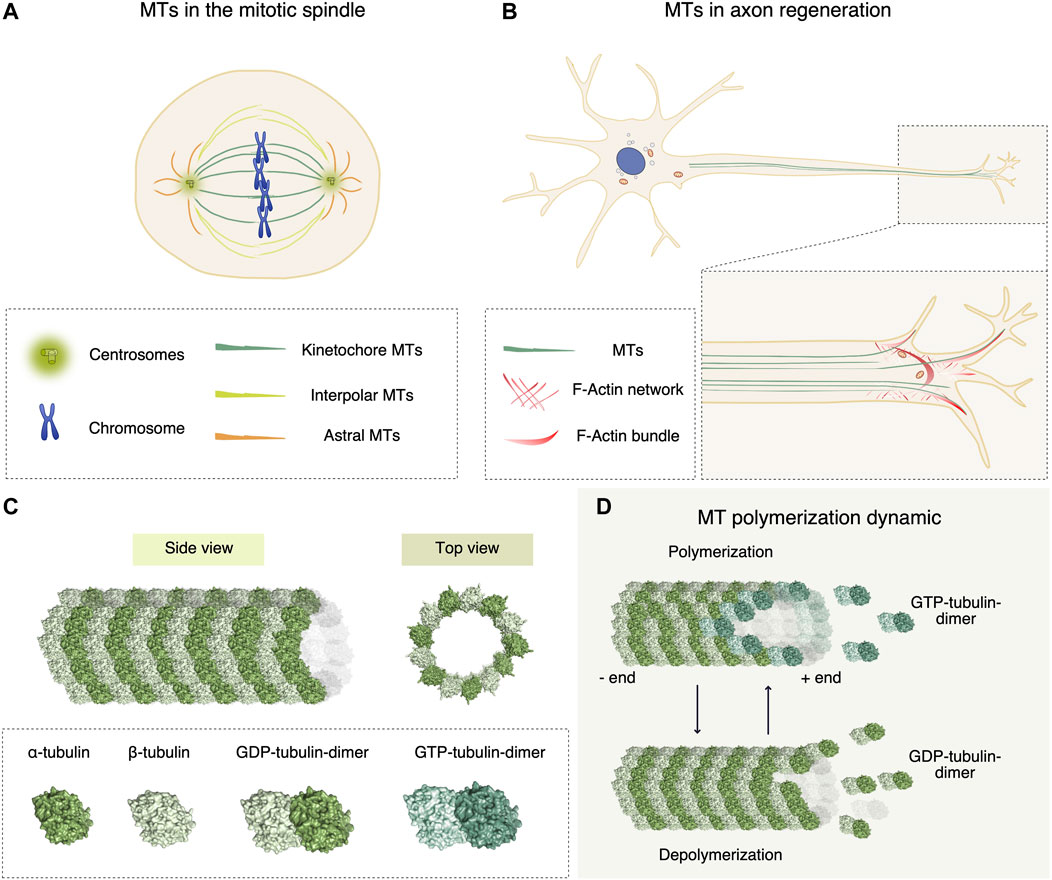
Nucleophilic Acyl Substitutions of Anhydrides with Protic Nucleophiles Catalyzed by Amphoteric, Oxomolybdenum Species | The Journal of Organic Chemistry
Efficient and Practical Procedure for the Esterification of the Free α-Car- boxylic Acid of Amino Acid Residues with β-(Trimet

PDF) Versatile selective α-carboxylic acid esterification of N-protected amino acids and peptides by alcalase

Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing) DOI:10.1039/C4NP00106K

Nucleophilic Acyl Substitutions of Anhydrides with Protic Nucleophiles Catalyzed by Amphoteric, Oxomolybdenum Species | The Journal of Organic Chemistry

Convenient Synthesis of N-Methylamino Acids Compatible with Fmoc Solid-Phase Peptide Synthesis | The Journal of Organic Chemistry

Characterization of novel heterocyclic compounds based on 4-aryl-4H-chromene scaffold as anticancer agents: Design, synthesis, antiprofilerative activity against resistant cancer cells, dual β-tubulin/c-Src inhibition, cell cycle arrest and apoptosis ...

Synthesis of Microcolin B, a Potent New Immunosuppressant Using an Efficient Mixed Imide Formation Reaction | The Journal of Organic Chemistry
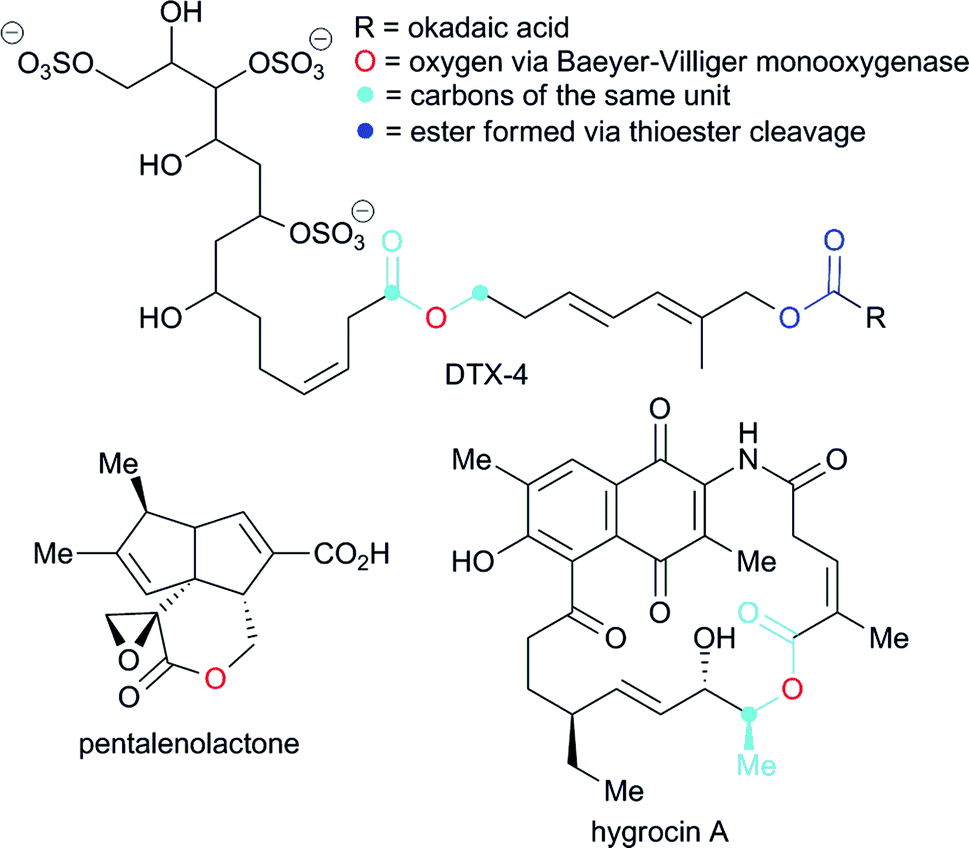
Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing) DOI:10.1039/C4NP00106K

PDF) Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products

The ester derivatives obtained by C-ring modification of podophyllotoxin induce apoptosis and inhibited proliferation in PC-3M cells via down-regulation of PI3K/Akt signaling pathway - ScienceDirect
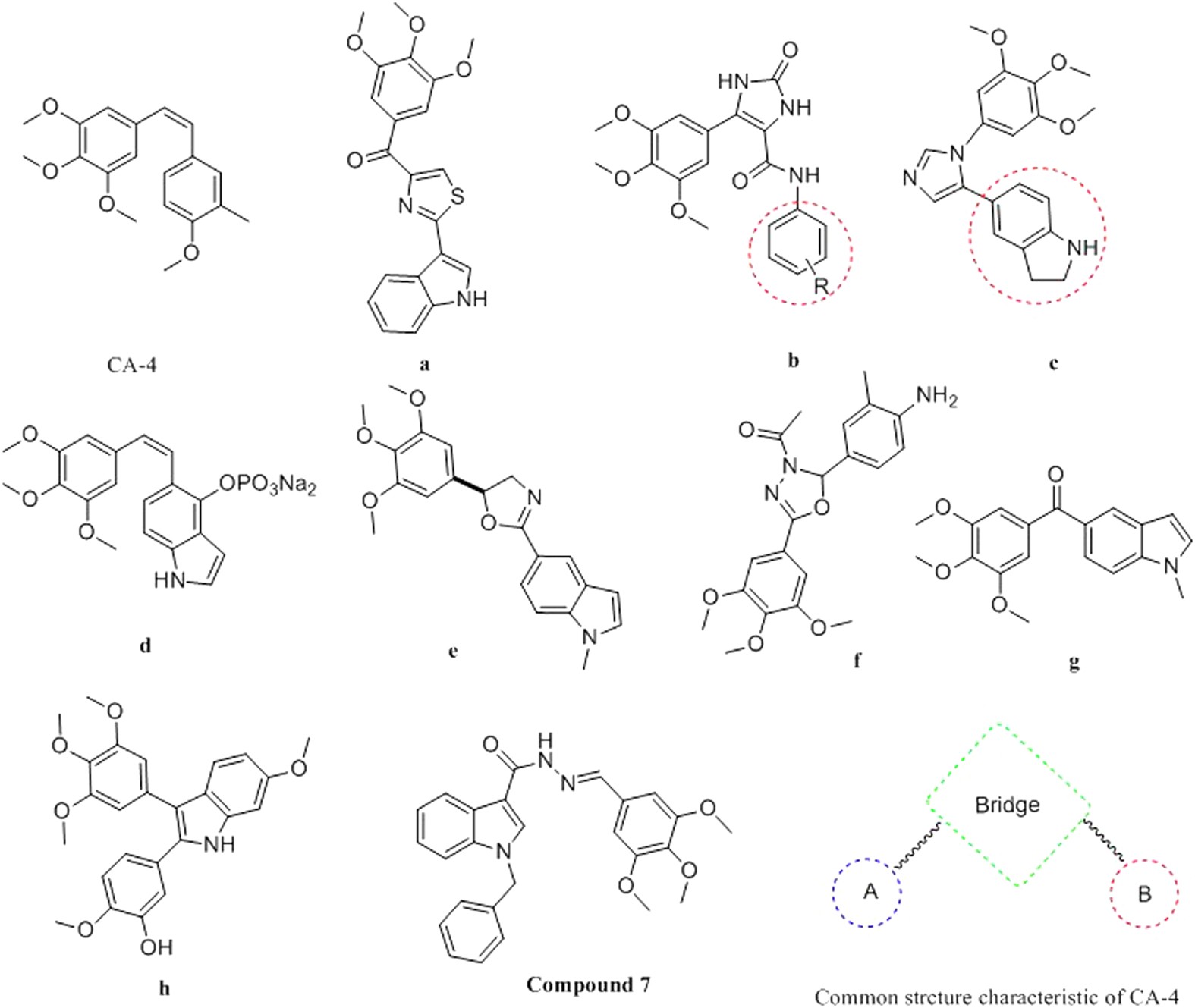
Design, Synthesis and Antitumor Activity of Novel link-bridge and B-Ring Modified Combretastatin A-4 (CA-4) Analogues as Potent Antitubulin Agents | Scientific Reports