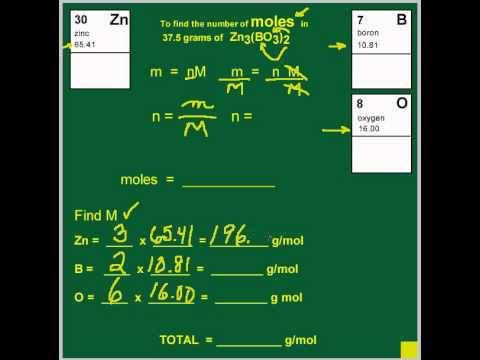
SOLVED: If 50.0 g of CH₃OH (MM = 32.04 g/mol) are added to a 500.0 mL volumetric flask, and water is added to fill the flask, what is the concentration of CH₃OH

SOLVED: If 25.0 g of KBr (MM = 119.00 g/mol) are added to a 500.0 mL volumetric flask, and water is added to fill the flask, what is the concentration of KBr

SOLVED: What is the concentration in molarity of a solution which is 11.39 %m/v octane (MM = 114.2 g/mol ) in hexane (MM = 86.18 g/mol)?

mm-GBSA binding energy (ΔG, kcal/mol) of molecules to (A) DNMT1 and (B)... | Download Scientific Diagram

PLEASE HELP QUICKLY!!! MM H2O2 = 34.02 g/mol MM H2O = 18.02 g/mol MM O2 = 32 g/mol 2H2O2 —> 2H2O + - Brainly.com
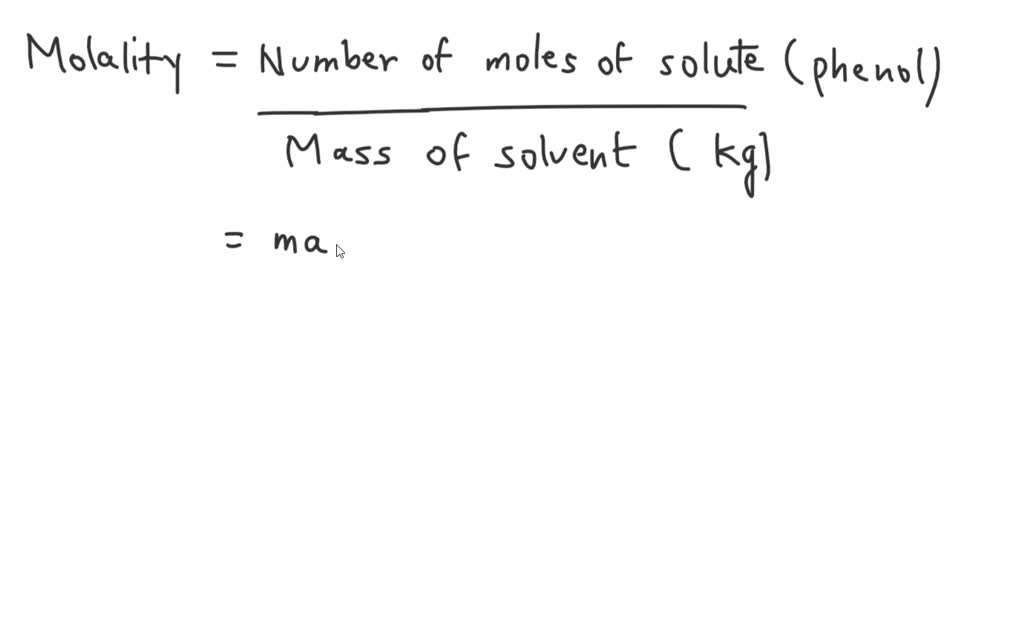
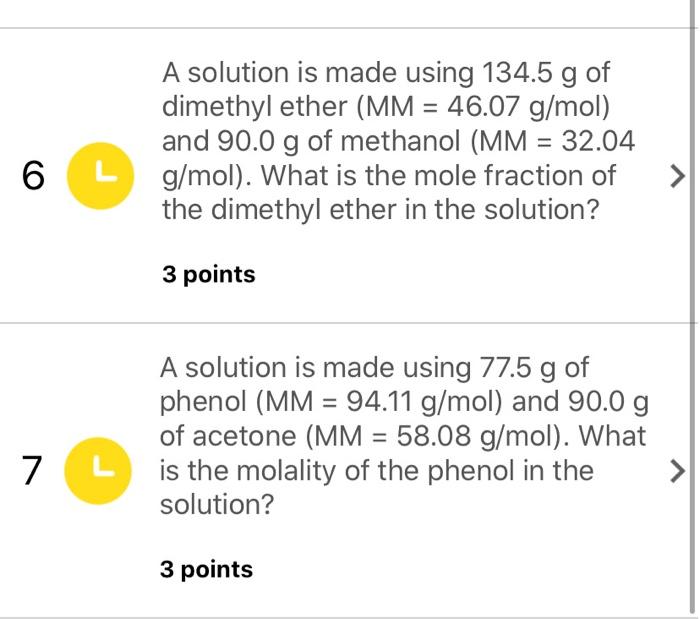
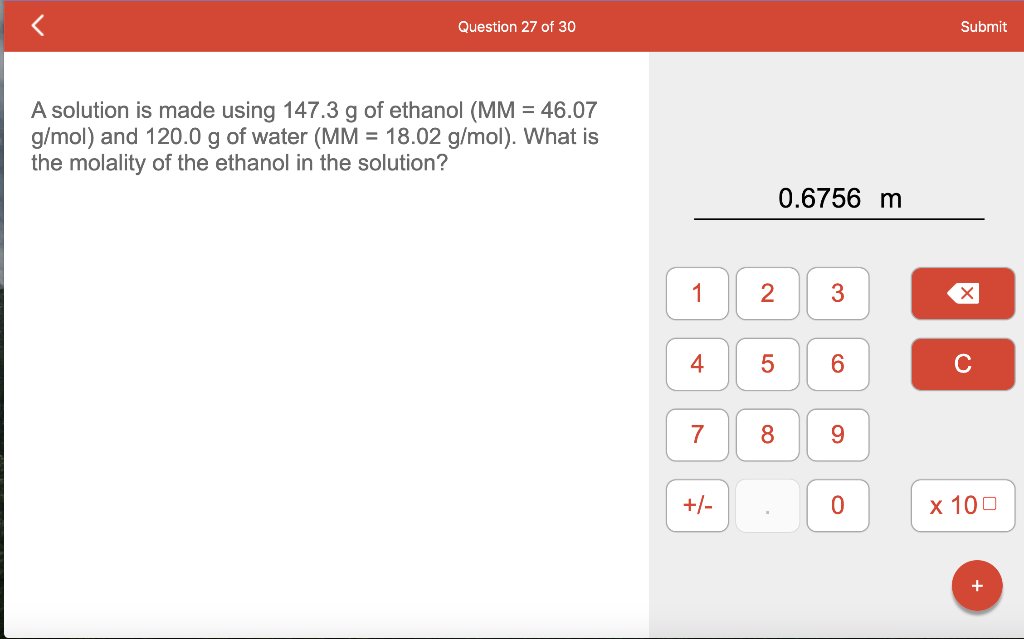
SOLVED: A solution is made using 79.9 g of phenol (MM = 94.11 g/mol) and 90.0 g of acetone (MM = 58.08 g/mol). What is the molality of the phenol in the solution?

The MM/GBSA ∆G bind and its energy components (kcal/mol). The ∆G bind,... | Download Scientific Diagram

Bahco Wrench 9029C BH9029C Adjustable MOL Central Gran AP Wrench, Silver/Black, 6 Inch, 32 mm - - Amazon.com