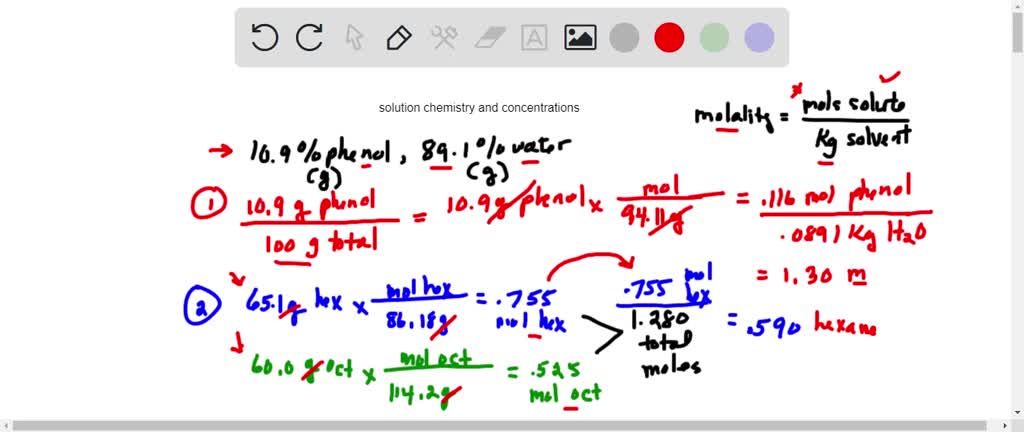
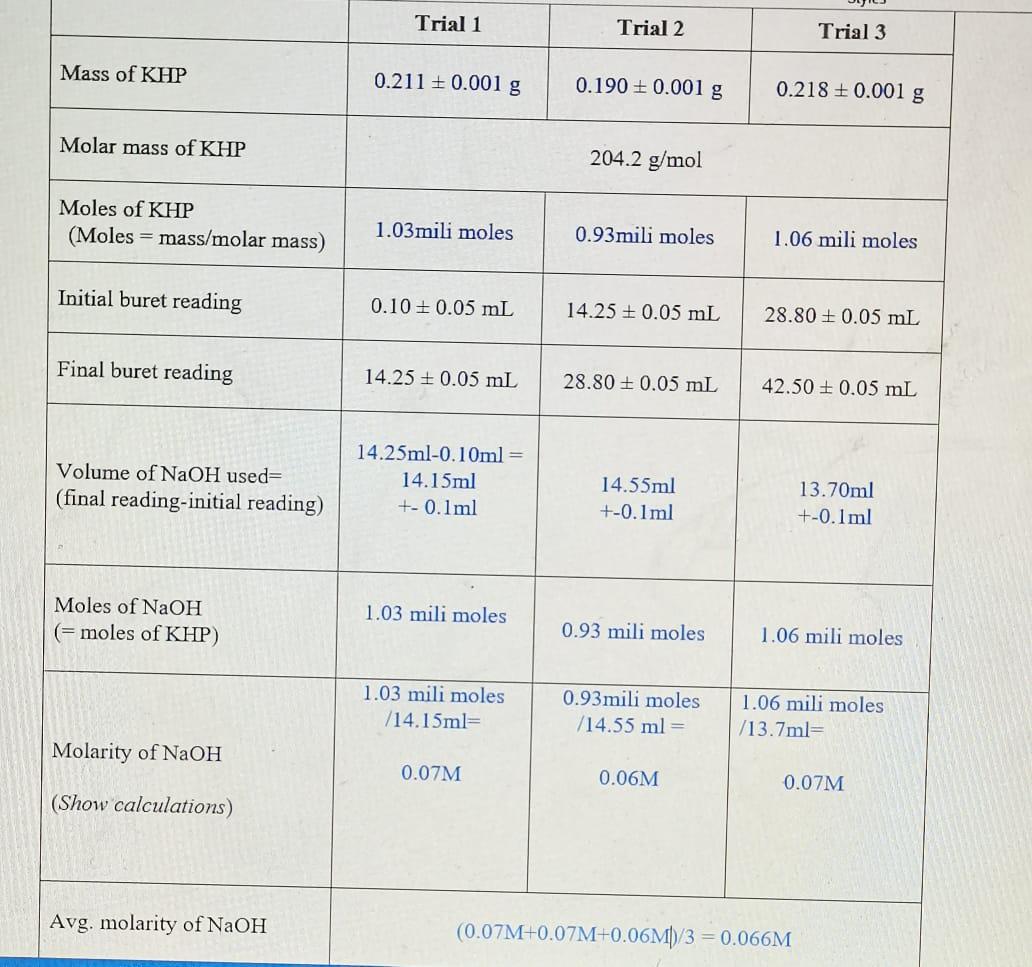
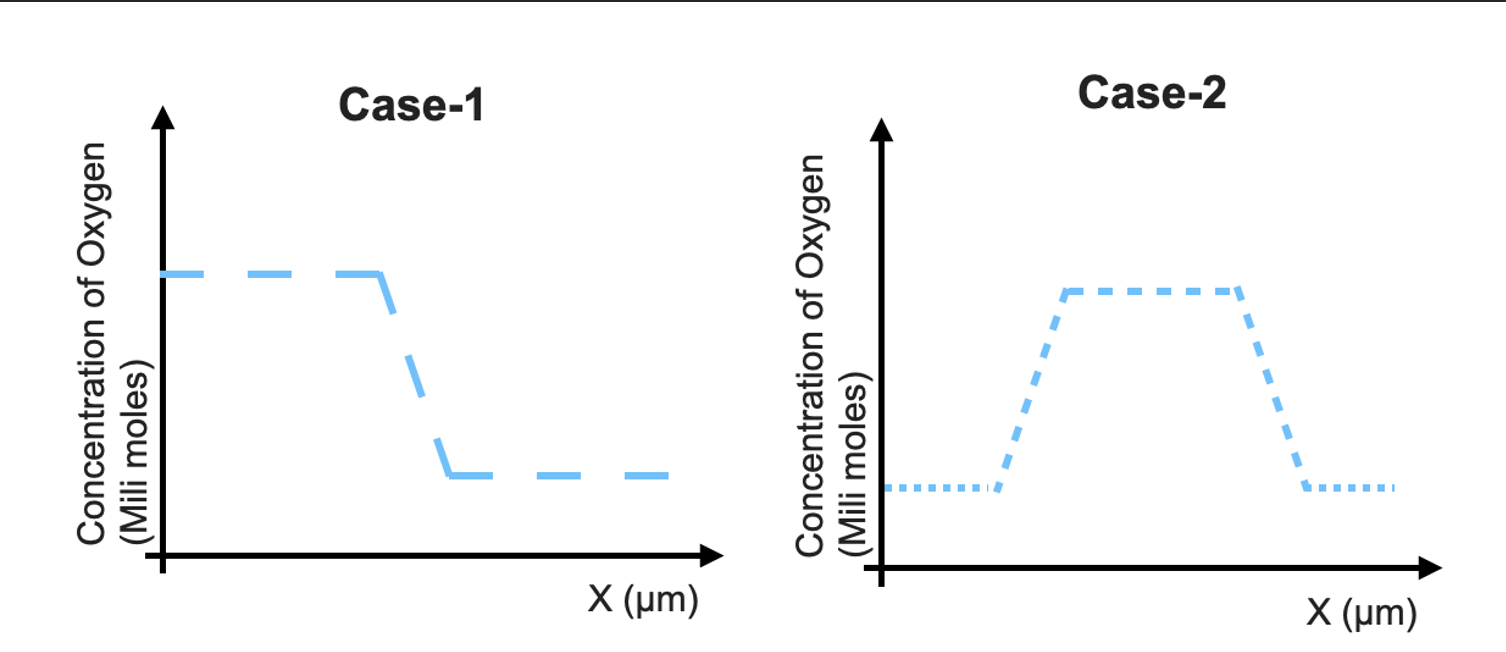
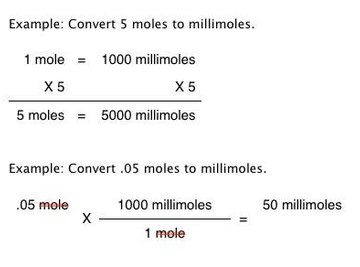
100 mL of 0.6 M acetic acid is shaken with 2 g activatid carbon . The final concetration solutio... - YouTube

8 Selected Books (Set) Of Bimal Mitra (Kharidi Kaudiyon Ke Mol - Vol 1-2, Begam Meri Biswas: Vol 1-2, Ve Aankhein,Sahab Bibi Gulam, Char Ankhon Ka Khel , Rokar Jo Mili Nahi):

How many milli moles of sucrose should be dissolved in 500 gms of water so that the difference between the boiling point and freezing point of the solution becomes 103.57^oC ? (Kf =

PDF) Pharmacological Basis for Abrogating Myocardial Reperfusion Injury Through a Multi-Target Combined Antioxidant Therapy: Supplementary material