How to arrange the following molecules or ions in the order of their increasing bond length N2, N2^-, and n2^2- - Quora
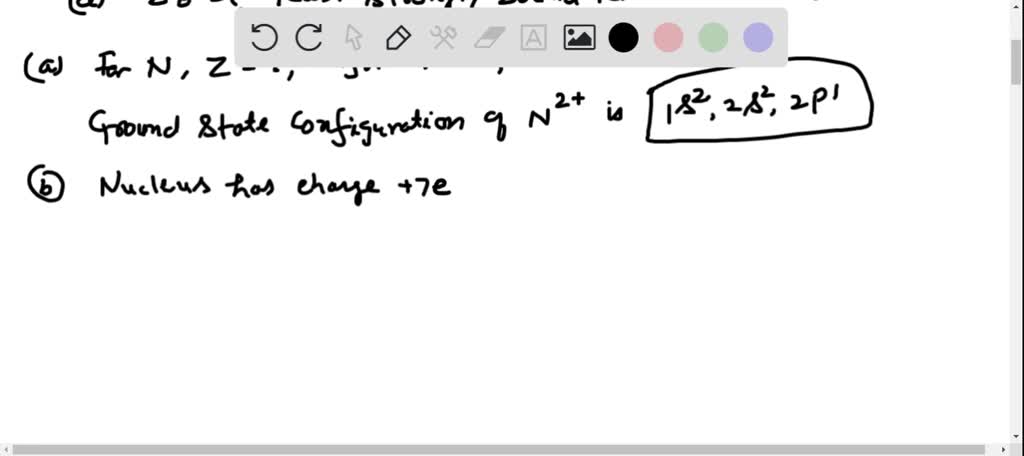
SOLVED:(a) The doubly charged ion N^2+ is formed by removing two electrons from a nitrogen atom. What is the ground-state electron configuration for the N^2+ ion? (b) Estimate the energy of the








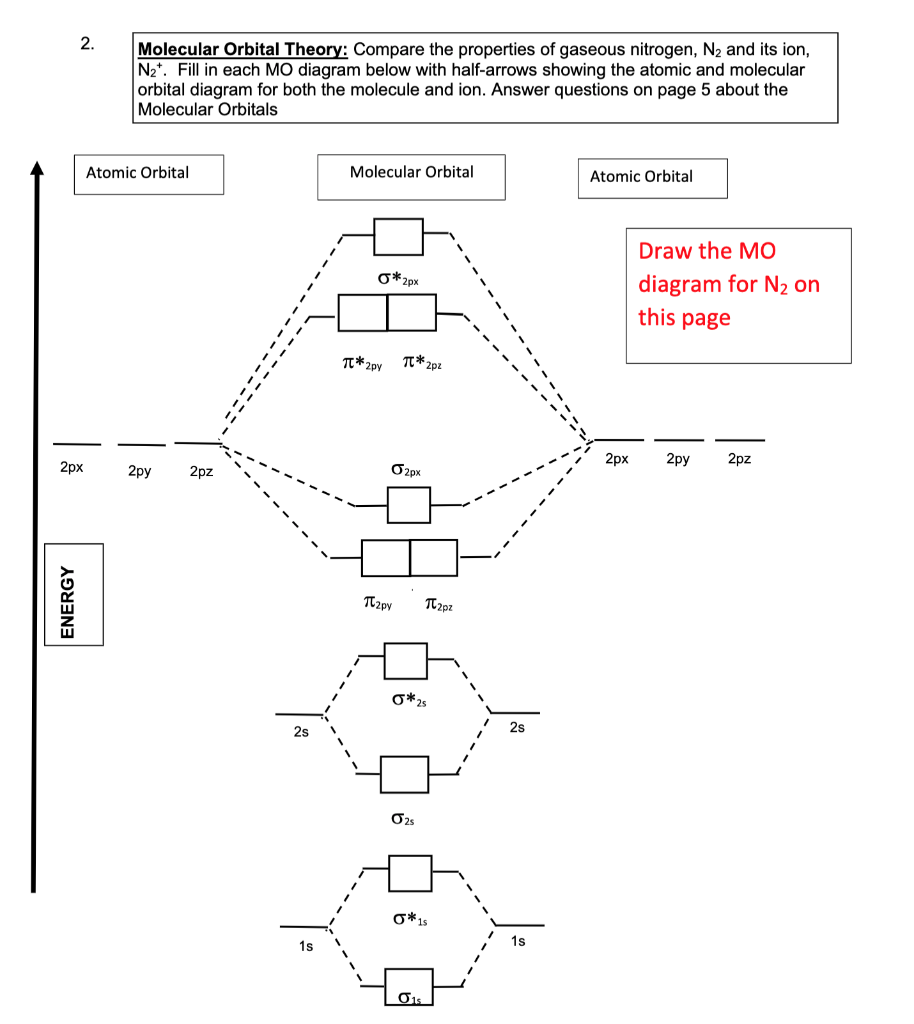

![The bond order of \\[{N_2}^ + \\] ion is:A.\\[1\\]B.\\[2\\]C.\\[2.5\\]D.\\[3\\] The bond order of \\[{N_2}^ + \\] ion is:A.\\[1\\]B.\\[2\\]C.\\[2.5\\]D.\\[3\\]](https://www.vedantu.com/question-sets/3d3b1779-d1e3-4026-b77d-c9d750797a82788781133640851459.png)