Reduction of O2 to H2O and its free radical intermediates (A) Lewis... | Download Scientific Diagram

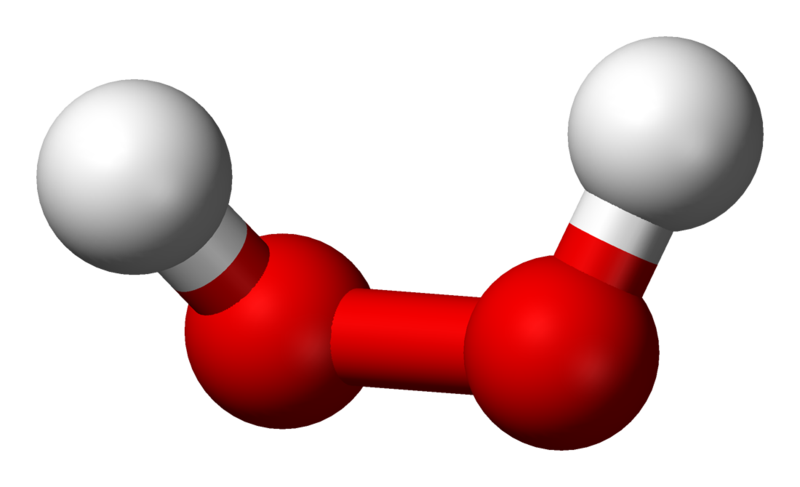
SOLVED: Balance the following equation: H2O2 HzO + 02 H202 HzO + 02 2 H2O2 2 HzO + 02 2 HzO2 2 H2O + 302 H2Oz 2 HzO + 02
, RE(SO4)[B(OH)4](H2O)2, and RE(SO4)[B(OH)4](H2O)·H2O: Rare-Earth Borate-Sulfates Featuring Three Types of Layered Structures | Inorganic Chemistry RE(SO4)[B(OH)4](H2O), RE(SO4)[B(OH)4](H2O)2, and RE(SO4)[B(OH)4](H2O)·H2O: Rare-Earth Borate-Sulfates Featuring Three Types of Layered Structures | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/acs.inorgchem.7b02317/asset/images/large/ic-2017-02317c_0008.jpeg)
RE(SO4)[B(OH)4](H2O), RE(SO4)[B(OH)4](H2O)2, and RE(SO4)[B(OH)4](H2O)·H2O: Rare-Earth Borate-Sulfates Featuring Three Types of Layered Structures | Inorganic Chemistry
I) H2O2 + O3 → H2O + 2O2 (II) H2O2 + Ag2O → 2Ag + H2O + O2 Role of hydrogen peroxide in the - Sarthaks eConnect | Largest Online Education Community

Kinetic studies on the reaction of 2 with H2O2 in buffered MeCN/H2O... | Download Scientific Diagram
1) H2O2 + O3 → H2O +2O2 2)H2O2 +Ag2O →2Ag +H2O +O2 Determine whether H2O2 is oxidised or reduced in the above reaction? Explain.






![Water [H2O] Molecular Weight Calculation - Laboratory Notes Water [H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/water-molecular-weight-calculation.jpg)