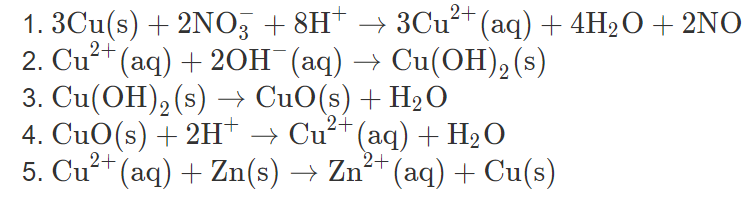Lab 21Date: Cupric Oxide from Cupric Sulfate Purpose Compare the experimental and calculated amounts of CuO produced in a rxn. Background CuSO 4 (aq) + - ppt download

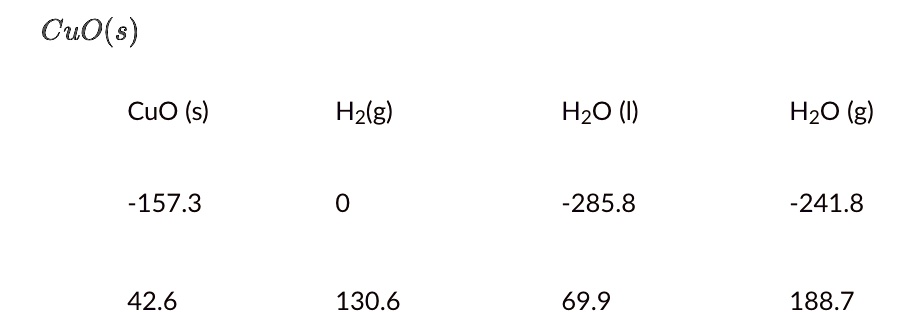
Given the redox reaction: CuO (s) + H2(g)harr Cu(s) + H2O(g) i) Identify the species which undergo reduction and which undergo oxidation

Combined Experimental and CFD Investigation of the Parabolic Shaped Solar Collector Utilizing Nanofluid (CuO-H2O and SiO2-H2O) as a Working Fluid

Los números correctos de oxidación para la siguiente reacción son: CuO + H2 → Cu + H2O O +2, -2, 0, 0, +1, - Brainly.lat
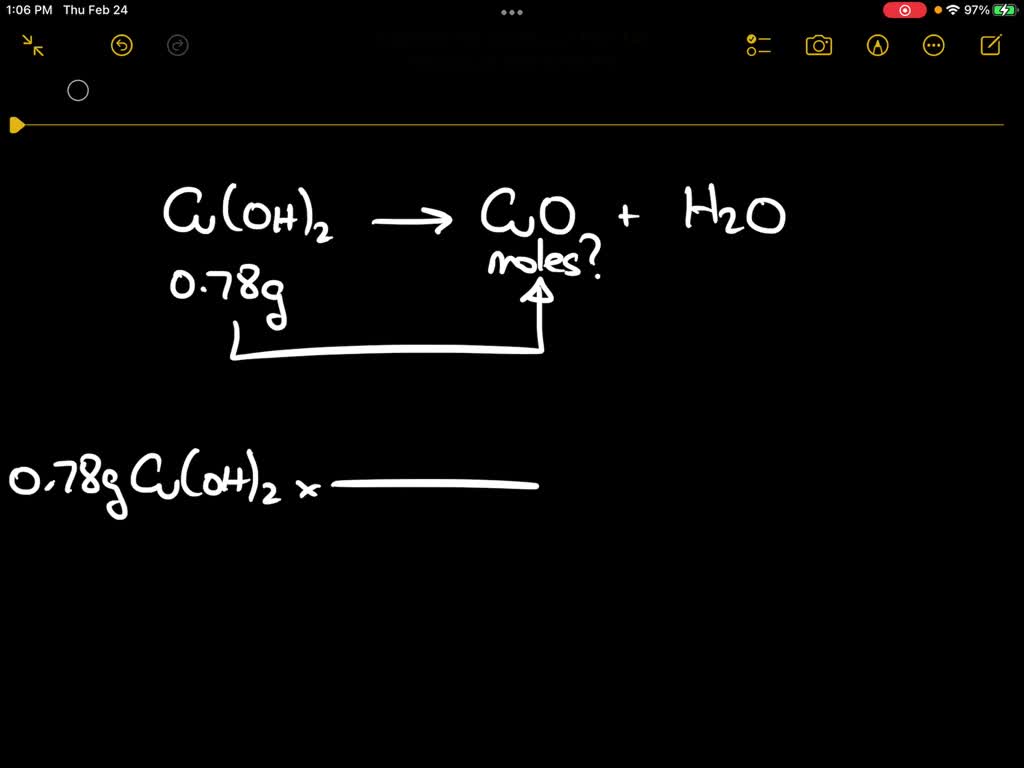
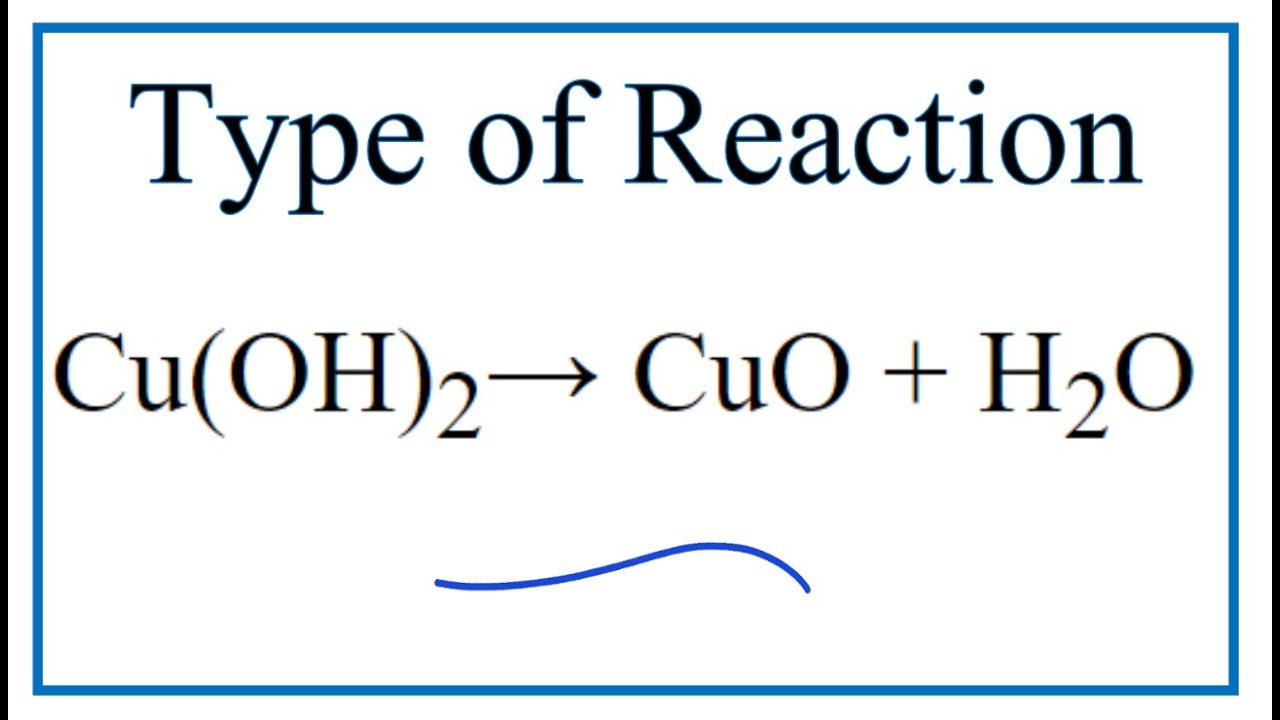
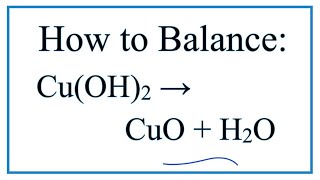
SOLVED: Upon heating Cu(OH)2 decomposes into CuO and H2O as shown below. Cu(OH)2 → CuO + H2O If 0.78 g of Cu(OH)2 was decomposed, calculate the moles of CuO produced Use 29Cu63.5,

Pushing the Limits of Rapid Anodic Growth of CuO/Cu(OH)2 Nanoneedles on Cu for the Methanol Oxidation Reaction: Anodization pH Is the Game Changer | ACS Applied Energy Materials