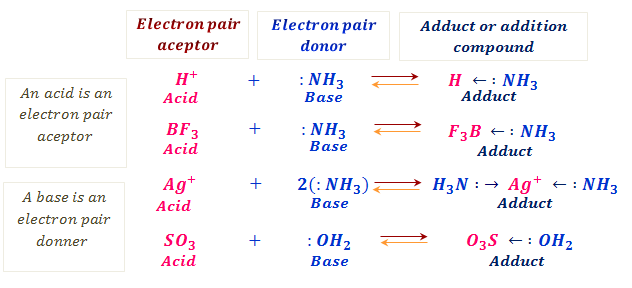
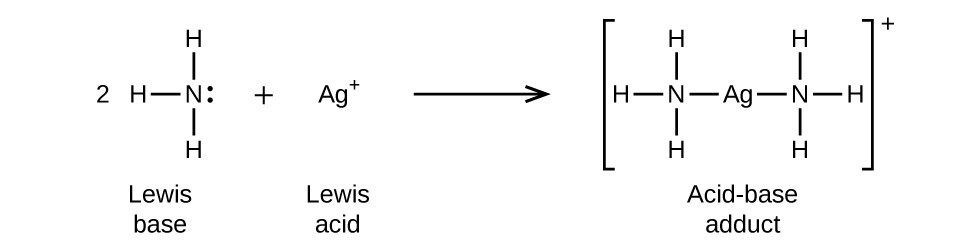
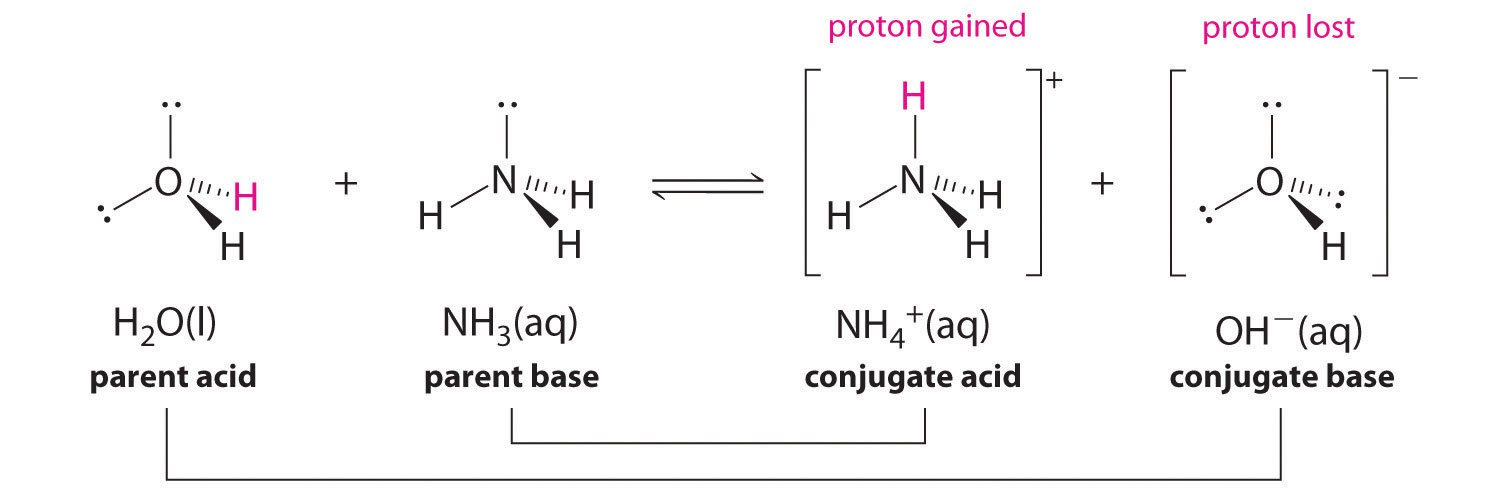
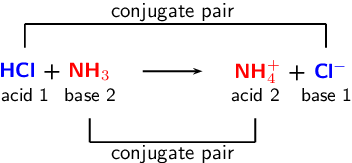
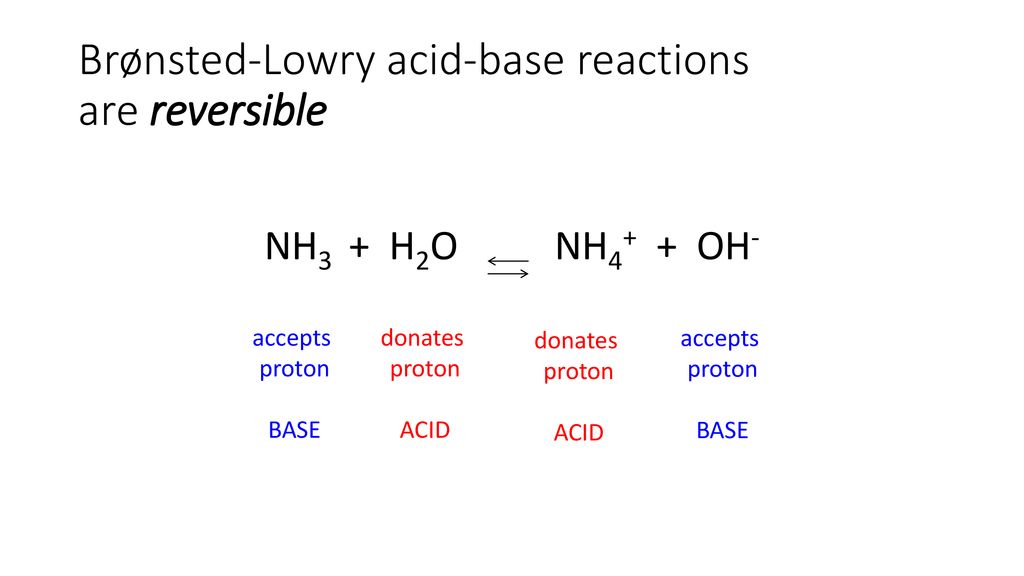
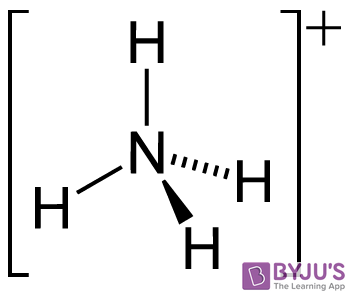
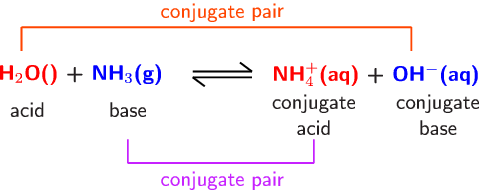
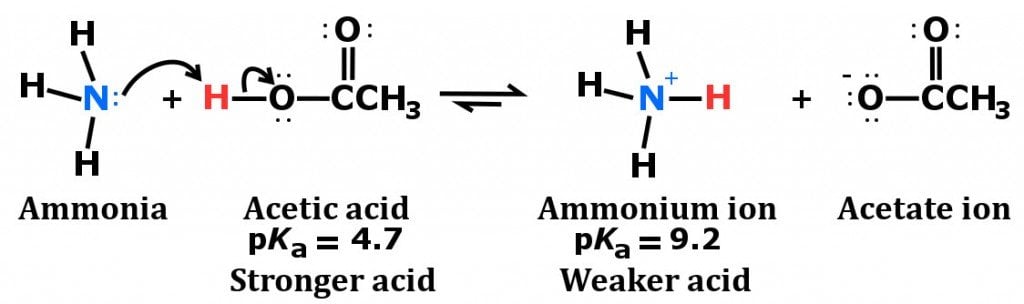
Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com

NH3 is a weak base (Kb = 1.8 times 10^-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.050 M in
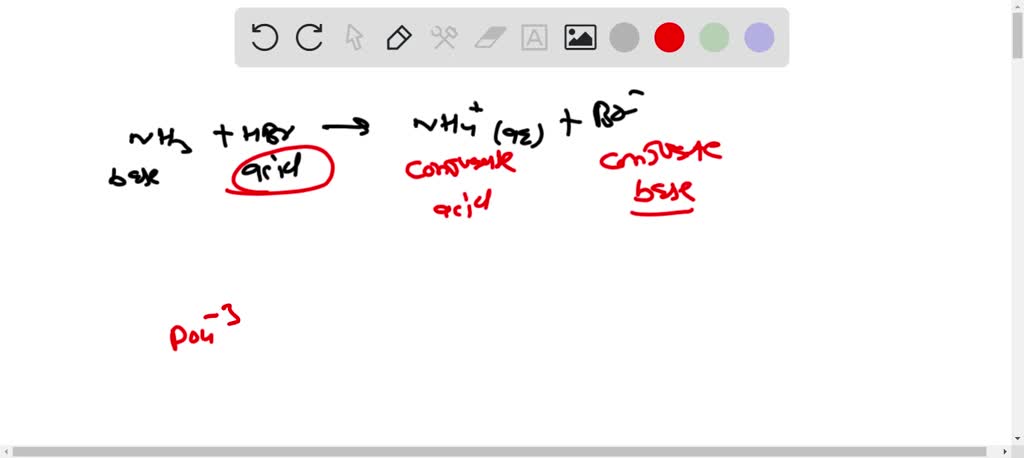
SOLVED: 16. Write an equation that shows the reaction of ammonia, NH3 with hydrobromic acid, HBr. Label the acid,the base, the conjugate acid, and the conjugate base. Write an equation that shows

Electric Field-Driven Acid−Base Chemistry: Proton Transfer from Acid (HCl) to Base (NH3/H2O) | The Journal of Physical Chemistry A