
Factory Price Disodium Phosphate Dihydrate Na2hpo4.2h2o - Buy Factory Price Disodium Phosphate Dihydrate,Na2hpo4.2h2o,Disodium Phosphate Dihydrate Factory Product on Alibaba.com
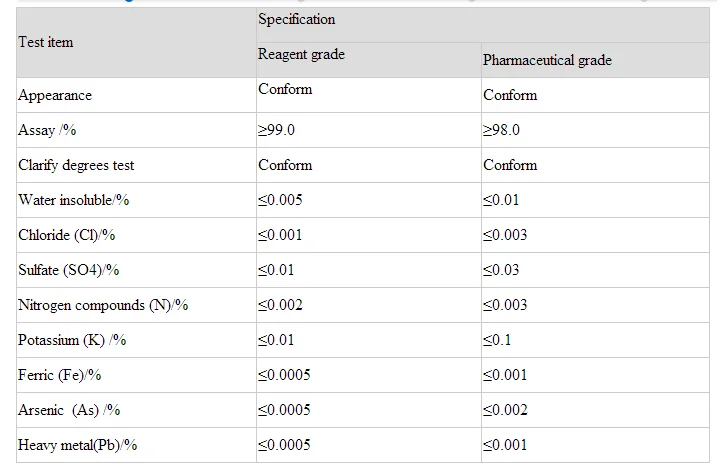
Factory Price Disodium Phosphate Dihydrate Na2hpo4.2h2o - Buy Factory Price Disodium Phosphate Dihydrate,Na2hpo4.2h2o,Disodium Phosphate Dihydrate Factory Product on Alibaba.com

Comparative Study of Sodium Phosphate and Sodium Sulfate in Aqueous Solutions at (298.15 to 353.15) K | Journal of Chemical & Engineering Data
Q. The equivalent mass of H3PO4 (Molecular weight = 98 g/mol) and Na2HPO4 (Molecular weight = 142 g/mol) in the reaction are respectively : H3PO4 + 2NaOH → Na2HPO4 + 2H2O (1) 49, 142 (2) 49, 71 (3) 98, 71 (4) 98, 142

Spectrum S1108-2.5KG (SPFDB) Sodium Phosphate, Dibasic, Dihydrate, FCC, Na2HPO4.2H2O: Amazon.com: Industrial & Scientific
SODIUM DIHYDROGEN PHOSPHATE STARTING FROM SODIUM CHLORIDE AND ORTHOPHOSPHORIC ACID VIA CATION RESIN EXCHANGE Doan Pham Minh, A

Spectrum S1108-12KG (SPFDB) Sodium Phosphate, Dibasic, Dihydrate, FCC, Na2HPO4.2H2O: Amazon.com: Industrial & Scientific

1 Bán Hóa chất di-Sodium hydrogen phosphate dihydrate, - Na2HPO4.2H2O - SO0339 - Scharlau giá rẻ ở hcm

Measurement and Modeling of the Solubility of NH4VO3 in the Na2HPO4–H2O and (NH4)2HPO4–H2O Systems | Journal of Chemical & Engineering Data

![Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/01/sodium-phosphate-dibasic-dihydrate-molecular-weight-calculation-300x186.jpg)
![di-sodium hydrogen phosphate | CAS [10028-24-7] | BIOpHORETICS™ di-sodium hydrogen phosphate | CAS [10028-24-7] | BIOpHORETICS™](https://biophoretics.com/879-panda_thickbox_default_2x/di-sodium-hydrogen-phosphate2h2o-cas-10028-24-7-serva.jpg)







.jpg)