Existence of a Hydroperoxy and Water (HO2·H2O) Radical Complex | The Journal of Physical Chemistry A
Shock tube study of the rate constants for H + O2 + M → HO2 + M (M = Ar, H2O, CO2, N2) at elevated pressur
![PDF] A 5-step reduced mechanism for combustion of CO/H2/H2O/CH4/CO2 mixtures with low hydrogen/methane and high H2O content | Semantic Scholar PDF] A 5-step reduced mechanism for combustion of CO/H2/H2O/CH4/CO2 mixtures with low hydrogen/methane and high H2O content | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cc5a382eb7f4df0f4efd57ed020729765916e685/17-FigureA.4-1.png)
PDF] A 5-step reduced mechanism for combustion of CO/H2/H2O/CH4/CO2 mixtures with low hydrogen/methane and high H2O content | Semantic Scholar

SOLVED:A possible mechanism for the decomposition of hydrogen peroxide is H2O2⟶ 2 OH H2O2+OH⟶H2O+HO2 HO2+OH⟶H2O+O2 Using your results from Exercise 39, specify which step is the rate-determining step. What is the overall

SOLVED: A proposed mechanism for the decomposition of hydrogen peroxide indludes the steps shown below. Whal islare the intermediale(s)? Step 1: H202 (8) + 2 OH (8) Step" 2: H202 (g) +
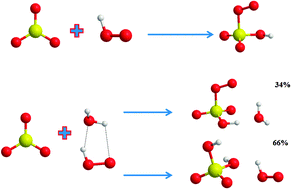
The reactions of SO3 with HO2 radical and H2O⋯HO2 radical complex. Theoretical study on the atmospheric formation of HSO5 and H2SO4 - Physical Chemistry Chemical Physics (RSC Publishing)

Theoretical studies on reactions of the stabilized H2COO with HO2 and the HO2···H2O complex. | Semantic Scholar

Atom Tunneling in the Water Formation Reaction H2 + OH → H2O + H on an Ice Surface | ACS Earth and Space Chemistry

Adsorption of Atmospherically Relevant Gases at the Air/Water Interface: Free Energy Profiles of Aqueous Solvation of N2, O2, O3, OH, H2O, HO2, and H2O2 | Semantic Scholar