
Nitrogen-doped carbon supported nanocobalt for the synthesis of functionalized triazines via oxidative cleavage of biomass derived vicinal diols as carbon synthons - ScienceDirect

Bioisosteres of the Phenyl Ring: Recent Strategic Applications in Lead Optimization and Drug Design | Journal of Medicinal Chemistry

A green and sustainable approach for the synthesis of 1,5-benzodiazepines and spirooxindoles in one-pot using a MIL-101(Cr) metal–organic framework as ... - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D1NJ03176G

Nickel(II)–NΛNΛO Pincer Type Complex-Catalyzed N-alkylation of Amines with Alcohols via the Hydrogen Autotransfer Reaction | The Journal of Organic Chemistry

Stepwise Mechanism for the Bromination of Arenes by a Hypervalent Iodine Reagent | The Journal of Organic Chemistry

Nitrogen-doped carbon supported nanocobalt for the synthesis of functionalized triazines via oxidative cleavage of biomass derived vicinal diols as carbon synthons - ScienceDirect

Stepwise Mechanism for the Bromination of Arenes by a Hypervalent Iodine Reagent | The Journal of Organic Chemistry

Molecularly Engineering Defective Basal Planes in Molybdenum Sulfide for the Direct Synthesis of Benzimidazoles by Reductive Coupling of Dinitroarenes with Aldehydes | JACS Au

Nitrogen-doped carbon supported nanocobalt for the synthesis of functionalized triazines via oxidative cleavage of biomass derived vicinal diols as carbon synthons - ScienceDirect

Nitrogen-doped carbon supported nanocobalt for the synthesis of functionalized triazines via oxidative cleavage of biomass derived vicinal diols as carbon synthons - ScienceDirect
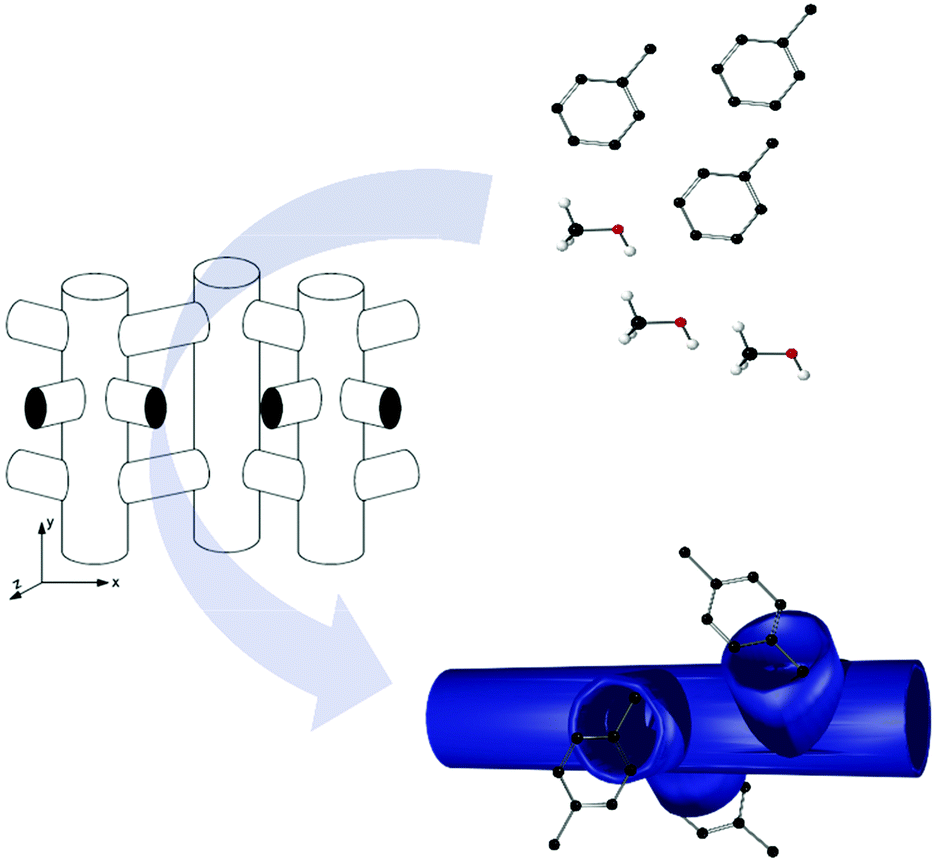
MOF-enabled confinement and related effects for chemical catalyst presentation and utilization - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D1CS00968K

Molecularly Engineering Defective Basal Planes in Molybdenum Sulfide for the Direct Synthesis of Benzimidazoles by Reductive Coupling of Dinitroarenes with Aldehydes | JACS Au






