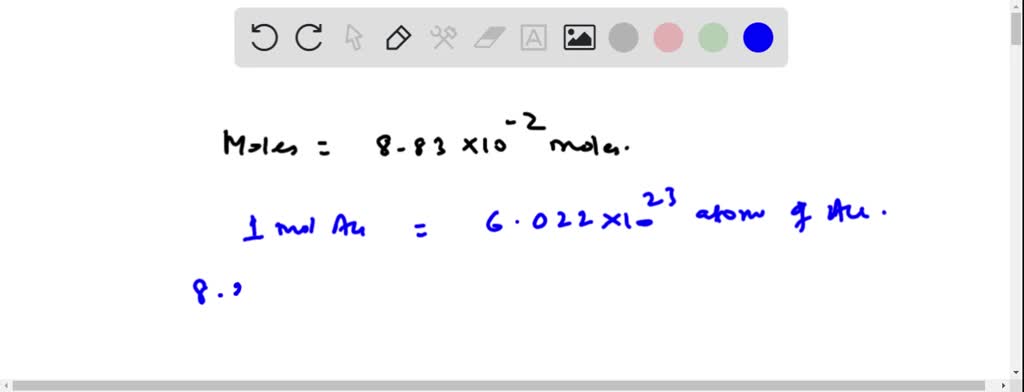
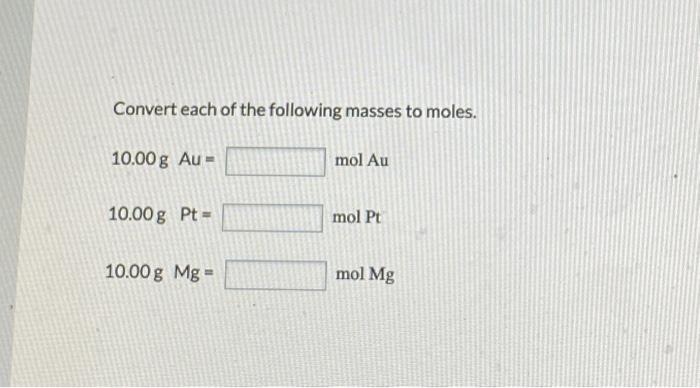
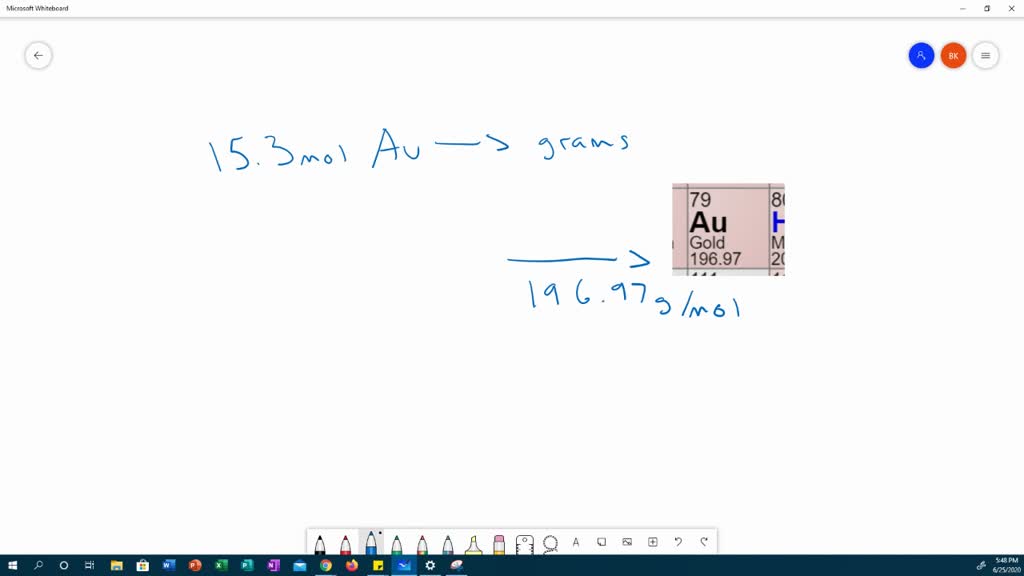
Figure S1. (A) CV of the Au electrode during the roughening process... | Download Scientific Diagram

Space / 404Potato on Twitter: "☕️ AU ...part 1? (maybe?) inspired by @MarvelWizardMom's idea #ironstrange #strangeiron #doctony https://t.co/gdacphGgz6" / Twitter

SOLVED: Convert these two atomic units (a.u.) into kJ/mol by using 2625 kJ/ mol = 1 a.u. -78.7939 kJ/mol -78.7838378 kJ/mol Please show the work.